Chemical Reactions
Properties
Uranium is a hard, dense, malleable, ductile, silver-white, radioactive metal of the actinide series in Group 3 of the periodic table. Uranium has three distinct forms. They are the orthorhombic crystalline structure, which is a highly reactive metal and reacts with almost all the non-metallic elements and their compounds. It dissolves readily in nitric and hydrochloric acids but resists attack by alkalies. It forms solid solutions and intermetallic compounds with many of the metals.
Uses of Uranium
The main uses of Uranium is for fuel in nuclear power reactors for electricity generation, in the manufacture of radioisotopes for medical applications and in nuclear science research using neutron fluxes.
The names, chemical formula and uses of two compounds of the element
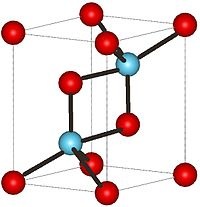
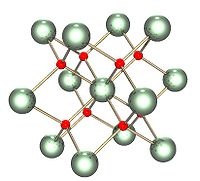
There are many names of Uranium in other languages but not in English. Two of the thirty seven compounds of Uranium, two are Uranium oxide and Uranium nitride. Uranium oxide is used for formulations of enamel, uranium glass, and porcelain. Uranium nitride as a potential fuel for generation IV reactors. Uranium Nitride is considered superior because of its higher fissionable density, thermal conductivity and melting temperature than the most common nuclear fuel, uranium oxide (UO2), while also demonstrating lower release of fission product gases and swelling, and decreased chemical reactivity with cladding materials. It also has a superior mechanical, thermal and radiation stability compared to standard metallic uranium fuel. The thermal conductivity is on the order of 4-8 times higher than that of uranium dioxide, the most commonly used nuclear fuel, at typical operating temperatures. Increased thermal conductivity results a lower thermal gradient between inner and outer sections of the fuel, potentially allowing for higher operating temperatures and reducing macroscopic restructuring of the fuel, which limits fuel lifetime.
Uranium is a hard, dense, malleable, ductile, silver-white, radioactive metal of the actinide series in Group 3 of the periodic table. Uranium has three distinct forms. They are the orthorhombic crystalline structure, which is a highly reactive metal and reacts with almost all the non-metallic elements and their compounds. It dissolves readily in nitric and hydrochloric acids but resists attack by alkalies. It forms solid solutions and intermetallic compounds with many of the metals.
Uses of Uranium
The main uses of Uranium is for fuel in nuclear power reactors for electricity generation, in the manufacture of radioisotopes for medical applications and in nuclear science research using neutron fluxes.
The names, chemical formula and uses of two compounds of the element
There are many names of Uranium in other languages but not in English. Two of the thirty seven compounds of Uranium, two are Uranium oxide and Uranium nitride. Uranium oxide is used for formulations of enamel, uranium glass, and porcelain. Uranium nitride as a potential fuel for generation IV reactors. Uranium Nitride is considered superior because of its higher fissionable density, thermal conductivity and melting temperature than the most common nuclear fuel, uranium oxide (UO2), while also demonstrating lower release of fission product gases and swelling, and decreased chemical reactivity with cladding materials. It also has a superior mechanical, thermal and radiation stability compared to standard metallic uranium fuel. The thermal conductivity is on the order of 4-8 times higher than that of uranium dioxide, the most commonly used nuclear fuel, at typical operating temperatures. Increased thermal conductivity results a lower thermal gradient between inner and outer sections of the fuel, potentially allowing for higher operating temperatures and reducing macroscopic restructuring of the fuel, which limits fuel lifetime.